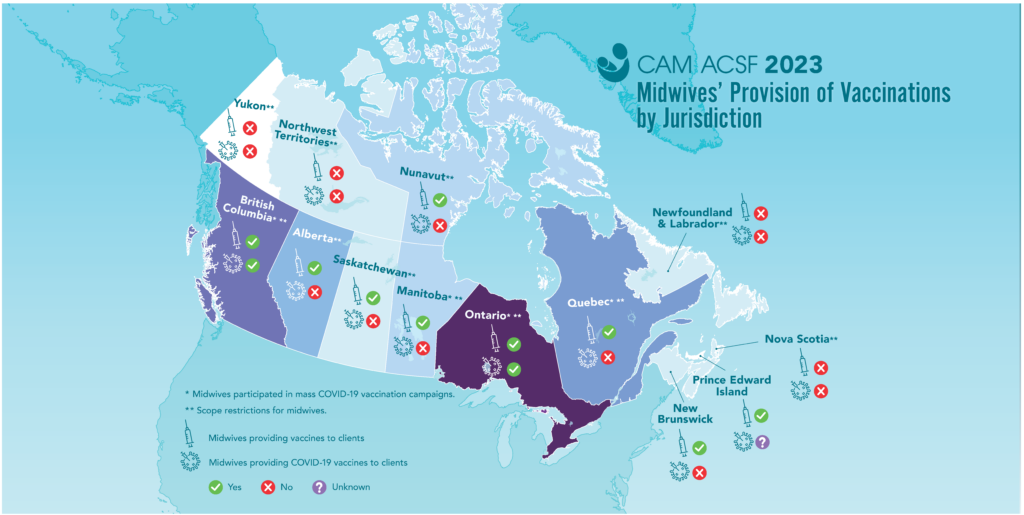
Access to vaccination information and services is a human right, and increasing safe and equitable access to vaccines and vaccine information is important for community health and well-being.
- Promoting Wellness Midwives advocate for normal, physiological pregnancy, birth, and the newborn period. Discussing interventions, including vaccination, is a component of this approach.
- Informed Choice Midwives are committed to respectfully delivering comprehensive, evidence-based immunization information and acknowledge client autonomy in decision making.
- Active Engagement Midwives welcome questions and discussion, promote active client participation in health care decision-making, strive to hear diverse perspectives, respond to a diversity of needs, and support open social discourse around vaccination.
4 New RSV Job Aids!